Lets talk lipid oxidisation
Whether you have heard of lipid oxidisation or not, you have probably done it before. Lipid oxidisation is what occurs when oils, also known as lipids are heated or reheated and go through a redox reaction called oxidisation. In more simple terms, science. There is a lot of information circling around about what ‘diet’ is best, and what’s most confusing is everyone has a different opinion on the subject. It’s very hip hop and happening to be substituting your olive oils with rapeseed oil, coconut oil or sunflower oil for cooking. But is it the better choice when it’s heated? What I’m going to focus on is based on research presented by Dr. Micheal Mosley, Trust me I’m a doctor; which oils are best to cook with?
One thing I have learnt during my time in Iceland is how popular it is to cook with oils such as sunflower or canola oil, so I was curious to understand why so many people cook with these products. The only time I hear Icelanders not talking about the weather is when they are talking about grilling. So let’s look into what’s going on when you throw a shrimp on the barbie.
What the heck is an aldehyde?
Aldehydes are found in many things, have multiple purposes and characteristically known for the smells they produce. They are used in plastics, perfumes, pharmaceuticals and considered an important part of caramelisation when cooking.
science direct
Aldehydes are also found as industrial or environmental pollutants and volatile aldehydes. Formaldehyde, acetaldehyde, and acrolein have the most significant effects on human health and are found in high concentrations in tobacco smoke and combustion engine exhaust.
The toxic aldehydes are a result of degradation of the fatty acids in oil, and although some are volatile, others remain after frying. That is why than be found in cooked food. As they are very reactive compounds they can react with proteins, hormones and enzymes in the organism and impede its correct functioning.
So are aldehydes good, or what’s the go?
In short, some are safe and some aren’t. for example we know that high concentrations of formaldehyde are found in tobacco smoke. We also know that smoking tobacco is carcinogenic. What scientists are now focusing more on is the molecular mechanisms of aldehydes in cooking to understand the potential health implications on humans. When we are measuring aldehydes, we are looking at which ones become toxic when heated both with and without food. Below I have inserted a graph that can be used as a guide to what oils are a better substitute.
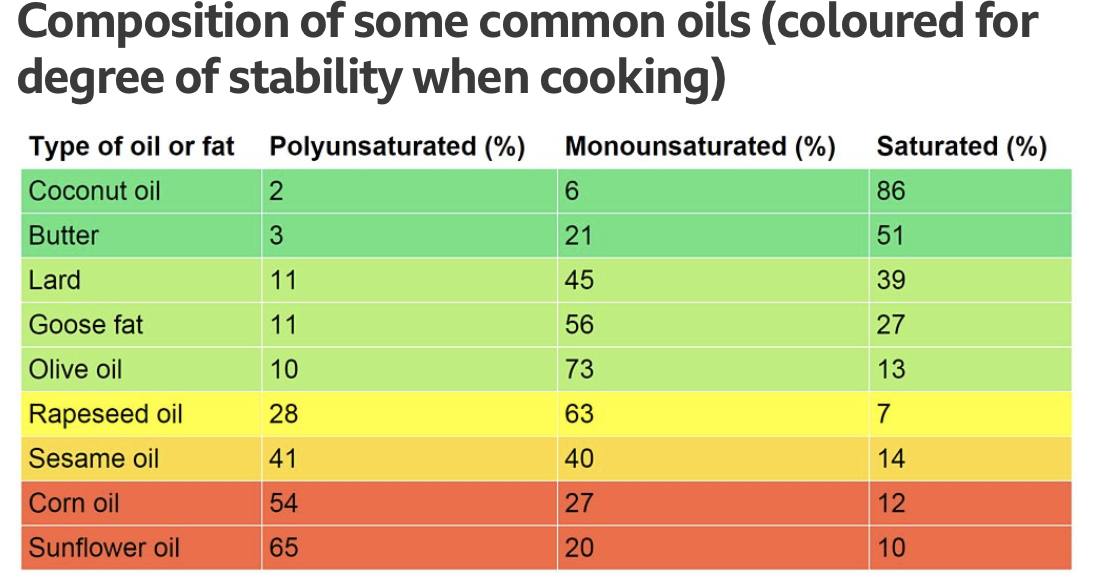
Recommendations
Michael Mosley suggests an olive oil to be a good overall substitute when considering poly and monounsaturated fats
Store your oils away from light and oxygen exposure, lids on ladies!
In general reducing oil intake is only going to better for overall health
Since the discovery of toxic aldehydes, oil companies are beginning to produce oil blends so those may be a better alternative when grilling
Links
http://presse.inra.fr/en/Press-releases/consumption-of-ultra-processed-food-and-risk-of-cardiovascular-disease
https://www.sciencedirect.com/science/article/pii/S1537189114000536
https://www.bbc.co.uk/programmes/articles/3t902pqt3C7nGN99hVRFc1y/which-oils-are-best-to-cook-with
https://onlinelibrary.wiley.com/doi/abs/10.1002/ejlt.201300273
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3436495/
https://books.google.is/books?hl=en&lr=&id=MeucDwAAQBAJ&oi=fnd&pg=PA211&dq=heated+oils+effects+on+health&ots=SeaXeIeApX&sig=CKalWnGTAQEeLoHVdyKafsg5SyQ&redir_esc=y#v=onepage&q=heated%20oils%20effects%20on%20health&f=false
https://www.researchgate.net/publication/248511136_Emissions_of_volatile_aldehydes_from_heated_cooking_oils
https://www.ncbi.nlm.nih.gov/pubmed/16417045
https://www.sciencedirect.com/topics/neuroscience/aldehydes
https://www.epa.gov/sites/production/files/2016-09/documents/acetaldehyde.pdf
https://socratic.org/questions/what-are-examples-of-aldehydes
https://www.toppr.com/guides/chemistry/aldehydes-ketones-carboxylic-acids/uses-of-aldehydes-and-ketones/
https://pubs.acs.org/doi/abs/10.1021/jf035241f
https://www.sciencedaily.com/releases/2012/02/120222093508.htm
https://link.springer.com/chapter/10.1007%2F978-1-4020-6704-4_11
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4106693/